I love learning about the chemistry of diseases, and about how microfluidics can be used to, firstly, better understand how they operate and, secondly, evaluate and refine potential therapeutic approaches that may become the foundation of a cure. To my delight, researchers from the Reches group at The Hebrew University of Jerusalem just published as an article entitled “Tailoring Peptide Coacervates for Advanced Biotechnological Applications: Enhancing Control, Encapsulation, and Antioxidant Properties” in ACS Applied Materials and Interfaces that looked at characterising the behaviour of coacervates (a form of chemical phase separation), how coacervates are part of many biolgical processes as well as a variety of neurodegenerative diseases (such as Alzheimer’s, Parkinson’s and Huntington’s diseases), and how their progress with coacervates could potentially apply to a variety of applications such as drug delivery.

So, what in blazes is a “coacervate”, and how does any of this relate to understanding and curing diseases, you ask? Great questions, the search for the answers to which led me to an excellent 2019 review in Annual Reviews entitled “Liquid–Liquid Phase Separation in Disease” by Alberti and Dormann at the Technical University of Dresden and Ludwig-Maximilians University in Munich, respectively. Since this part is really interesting and provides context for the relevance of the Israeli research, we’ll start here. Fair warning: this is rather complicated, but I’ll try to simplify it as much as possible, without misrepresenting anything (apologies to those in the field who detect an unwitting gaffe :-).
Liquid-liquid phase separation (LLPS) is a process by which one or several solutes in a solution self-concentrate into their own phase, essentially forming microdroplets termed condensates or coacervates. It turns out that LLPS is now believed to be fairly widespread for a number of different peptides and proteins within cells, and to underlie several biological processes; in some cases, these coacervates are essentially membraneless organelles in the cell. The review illustrates the general mechanisms by which abnormal LLPS may cause disease, such as a) genetic changes in a protein that change its solubility and thus tendency to form a coacervate, b) genetic changes in a regulator that changes when or where the coacervates forms (i.e. in the cytoplasm vs. nucleoplasm), and c) changes in physiological conditions (e.g. pH, salinity, osmotic pressure, etc.) that may promote or inhibit coacervate formation. The authors then provide generalised and specific examples of these mechanisms in action for several neurodegenerative diseases (such as Alzheimer’s, Parkinson’s and Huntington’s disease, frontotemporal dementia (FTD), amyotrophic lateral sclerosis (ALS or Lou Gehrig’s disease)), cancers (apparently many or all cancers via certain common processes such as high cellular replication) and infectious diseases (such as a large number of viruses via the formation of their coacervate viral factories, as well as many bacterial and fungal infections via common processes such as solidified cytoplasm in drug-resistant strains). Their Figure 3, reproduced above right as Figure i, is a helpful illustration of these processes happening in a cell.
Specific examples of these mechanisms are beyond the scope of this post (… and beyond my understanding of the biochemistry, if I’m being honest :-), but I find even the generalised examples fascinating, and maybe you will too. Paraphrasing directly from their caption for the figure above, here are some. In neurodegenerative diseases, mutations, abnormal posttranslational modifications (PTMs, normally used to complete the synthesis of a protein), altered subcellular localization (i.e. getting the proteins to the wrong part of the cell), or impaired protein quality control (PQC; an enzyme system that might e.g. keep a transported protein from phase-separating and losing function) can promote the formation of ectopic condensates (phase separation in the wrong cellular location) as well as precipitating to a solid, both leading to protein aggregates in diseased tissue. These aggregates can impair ribonucleoprotein (RNP) granules and cellular factors (proteins), contributing to neuronal dysfunction and cell death. In cancer, mutations in signaling receptors (e.g. nicotinic acetylcholine receptor, used to contract muscles in response to acetylcholine, or release dopamine in response to nicotine) can alter the formation of signalling clusters at sites of DNA transcription (into RNA) or DNA damage repair. This, in turn, can alter cellular signaling cascades, impair transcription or DNA damage repair, and thus promote a proliferative and malignant state of a cell (tumour growth). In viral infections, liquid-like viral factories form through phase separation of certain viral proteins, promoting viral genome replication or altering the antiviral immune response. Some antiviral sensors in our immune system detect “pathogen-associated molecular patterns” (PAMPs) and form a separate phase through LLPS upon binding to foreign DNA or RNA, thus stimulating an immune response.
So, why do any of these LLPS-based mechanisms matter? Well, the better they are understood, the better drugs and other therapeutic strategies can be designed to intercede and either hinder a diseases-based LLPS process, or promote/defend a natural one, to inhibit disease progress in the end. If we consider neurodegenerative diseases for example, it has been known for some time that they all show accumulations of cytoplasmic or nuclear proteins in certain regions of the brain, and that these protein aggregations are thought to drive the degeneration. The mechanisms by which this happens are fascinating, and are based on liquid-liquid phase separation.

In the case of ALS and frontotemporal dimentia (FTD), a key protein is “Fused in Sarcoma”, or FUS. FUS is involved in DNA transcription, repair and splicing as well as mRNA transport in cells such as neurons; in a 2013 paper entitled “Fused in sarcoma (FUS): An oncogene goes awry in neurodegeneration” published in Molecular and Cellular Neuroscience by Dormann, FUS’s role in neurodegeneration is described in detail, and its normal functions in the cell are illustrated in Figure 3 from the paper, reproduced at right as Figure ii. One failure mode is not fully performing the PTM of methylation of the arginine amino acids in the FUS protein. In FTD patients, the arginines are either unmethylated or monomethylated, which causes phase separation of the FUS, impairing its ability to act. A second failure mode involves mislocalisation of the FUS protein.
In the case of ALS, a mutation in FUS’s “nuclear localisation sequence” (NLS, the amino acid sequence ‘code’ along a protein like FUS that allows it to be imported into the cellular nucleus) impedes its ability to bind to nuclear import receptor Transportin (like an ‘admission pass’, activated by the NLS code, that allows the bearer (FUS) to enter the nucleus). Transportin not only clears its cargo protein (FUS or others) for entry into the nucleus, it also suppresses LLPS and aggregation, so impeded FUS-transportin binding leads to accumulation, phase separation and precipitation of the FUS in the cytoplasm (termed mislocalisation). In the case of FTD patients, the issue is aggregation of the Transportin itself, rendering it inactive, i.e. unable to bind to the FUS and chaperone it to the nucleus, leading again to FUS mislocalisation. Critically, these LLPS mechanisms suggest research and development opportunities in e.g. drug therapies that may block or counteract a particular disease mechanism such as genetic mutation, mislocalisation, etc.
These disease examples are the tip of the iceberg in this field of research; many more specific examples are laid out in Alberti and Dormann’s review. If the (hopefully accurate) summaries above piqued your interest, reading the full review will certainly provide a more fulsome understanding.
So, the relevance of LLPS chemistry to the modus operandi of neurodegenerative, cancer and viral diseases seems sound, but what benefit can microfluidic devices and processes confer? The answer to this is provided in the research article from Reches’ group. In a nutshell, they did a fairly thorough job of characterising synthetic coacervates made from short polypeptides, and looking at their ability to encapsulate and protect molecules within (e.g. for drug delivery).
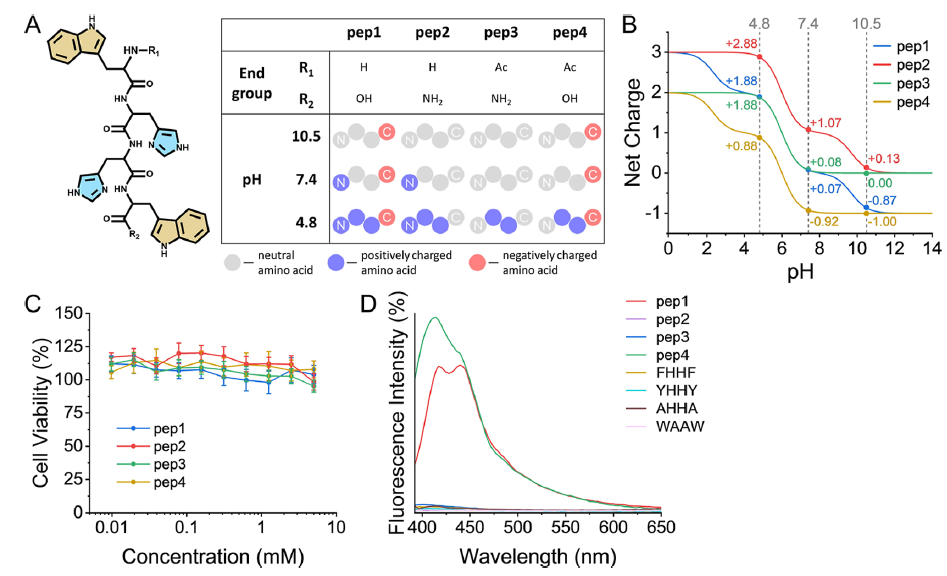
The researchers designed their four synthetic coacervates as being made from four short tetrapeptides, made from the amino acids tryptophan-(histidine)2-tryptophan, with different hydroxy, amino and acetyl terminations differentiating them. Their structure, charge states and biocompatibility were clearly shown in Figure 1 of their paper, reproduced at right as Figure iii. As a result of these different terminal groups, the four tetrapeptides, depicted in A, have significantly different pKa values at their acidic/basic groups, lending them different charges at a given pH, as shown in B. All four peptides performed well in ovarian cell viability tests (as a measure of biocompatibility), as shown in C.
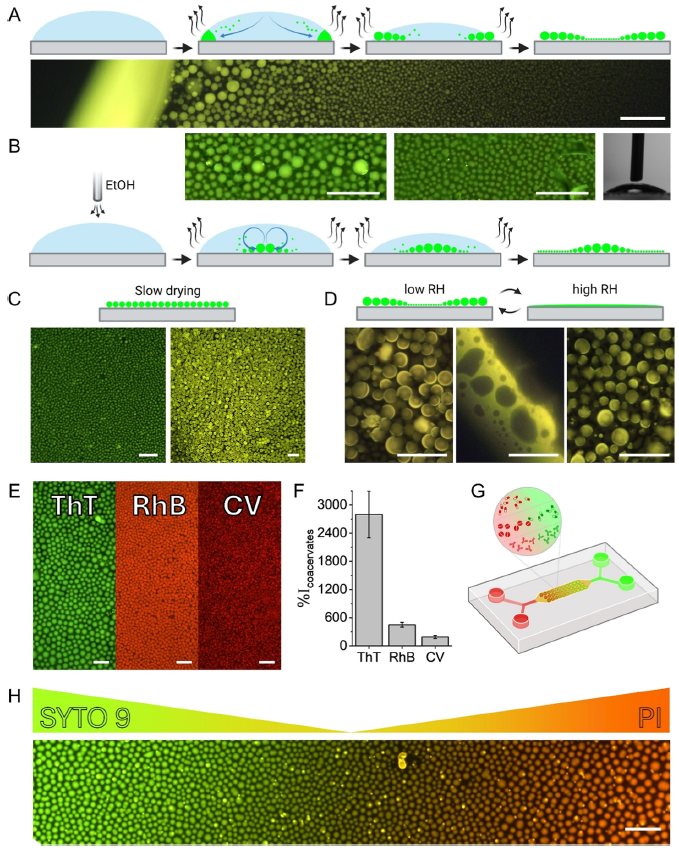
Characterisation of the formation and deployment of the peptide coacervate separated phase is well illustrated in Figure 6 of their paper, reproduced at right as Figure iv. In A, we see the normal evaporative formation of coacervate spheres (green) in a pH buffer solution; the smaller coacervate spheres are drawn to the solution drop perimeter by capillary action where they coalesce into larger droplets. The photo shows a fluorescence photomicrograph at the edge of the drop, with larger coacervate spheres towards the perimeter. The same process occurs in B, but ethanol vapour is blown over top of the solution drop to reduce the surface tension and in turn counter the capillary flow via the Marangoni effect (e.g. tears of wine), reversing the size distribution in the coacervate spheres vs. A. In C, slow drying is seen to produce a single coacervate size (or tight distribution, while in D, high relative humidity is shown to disassemble or merge the coacervate spheres (reversible with low RH). Frame E shows encapsulation of three dyes, thioflavin T, rhodamine B and crystal violet, in the coacervate spheres, while frame F shows the increase in fluorescence intensity for dye buffer solutions with the coacervate peptide vs. without (from ~2x to ~28x), showing the effective partitioning of the dyes in the coacervate phase. Frame G shows a schematic of the microfluidic device used to create dye gradients with green Syto 9 and red propidium iodide dyes, while frame H shows a photomicrograph of dye concentration gradient in the encapsulated dyes created by manipulation in the device. A somewhat closer look by the group at quantitative dye encapsulation showed a pH dependence, which is not surprising given the multiple acid groups and pKa values involved for each of the four tetrapeptide coacervates (referred to in Figure iii).
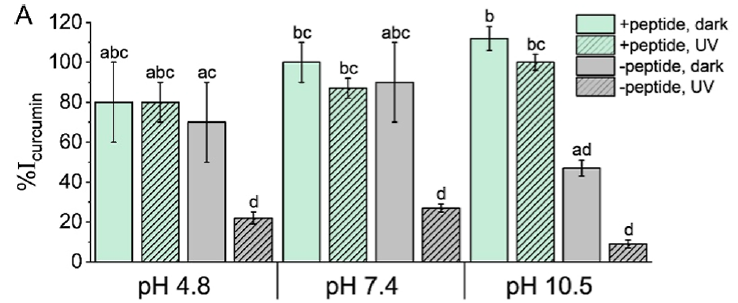
A last important area of performance they characterised was the protective antioxidant properties of the peptide coacervates on their encapsulated charge. They chose the popular dietary supplement curcumin — the natural polyphenol in turmeric giving it its yellow colour — as the encapsulated subject to test one of the coacervate’s protective abilities. Both alkaline pH and UV irradiation cause curcumin degradation and decomposition, so optical absorbance of intact curcumin was used to measure antioxidant protection afforded by the coacervate phase in comparison to a solution with no coacervate. The results are shown in their Figure 7A, shown at right as Figure v. At an acidic pH of 4.8 or neutral pH of 7.4, the unprotected curcumin (grey) was not significantly worse than the coacervate-protected curcumin (green), though the UV radiation did significantly degrade the unprotected curcumin (grey, hatched), but not the protected curcumin (green, hatched). At the alkaline pH of 10.5, both the alkalinity and UV radiation significantly degraded the unprotected curcumin (grey & grey hatched), but the coacervate was an effective protective sheath for the curcumin (green & green hatched).
If you’ve followed this far, then congratulations are in order 😉 , and let’s now consider what all this research might eventually translate into! First, let’s list some of the salient potential benefits of the synthetic tetrapeptide coacervates in the second paper:
- they’re made from amino acids, and are biocompatible in ovarian cells;
- they effectively encapsulate and concentrate the dyes tested;
- they confer alkaline and UV antioxidant protection;
- their sphere formation morphology can be tailored by several factors including humidity, ionic strength, presence of solvents (and perhaps surfactants?); and
- while these first peptide coacervates are a starting point, the nature of the coacervate phase could easily be tailored through choice of peptides (or other molecules or polymers), chain length, derivatisation, etc. to optimise host/drug compatibility, affinity or selectivity, encapsulation protection, etc.
How could this knowledge be built on? The authors speculated that the coacervate characterisation and performance shown in this study could lead to bio-applications such as the development biosensors as well as the encapsulated delivery of drugs and dietary supplements. I would add that there could potentially be organ-on-chip (OOC) applications, where the interactions of synthetic coacervates with the cells of a given target disease tissue, and perhaps with the natural coacervates within, could be tested. In some cases, such OOC studies would be precursors to the development of therapeutic drug delivery systems.
The authors made an initial foray into microfluidic manipulations to generate dye gradients. It is tempting to look further at the use of microfluidics to validate the effectiveness of coacervate constituents (peptides or others) to encapsulate a drug (or other ‘cargo’ molecule) under appropriate conditions, or the effectiveness of a coacervate-drug package to be delivered to the target organ tissue cells under the physiological conditions appropriate for that organ. Just as the dye gradient was shown here, other biologically relevant gradients could be explored. For coacervate encapsulation, perhaps pH, ionic strength, coacervate and drug concentrations, etc. would be of interest; for drug delivery, perhaps drug dosage, variations in physiological conditions owing to the condition itself, or owing to an additional condition — physiological changes at the cellular level in an Alzheimer’s patient that also has high cholesterol or diabetes, for example (I’m just musing about what particulars would be relevant). The ability to easily create gradients, isolate test areas, plumb reagents in and out, multiplex the conditions explored and tightly control processes on a small scale in microfluidics can help accelerate the development of a disease therapy built on the foundation of this knowledge.
